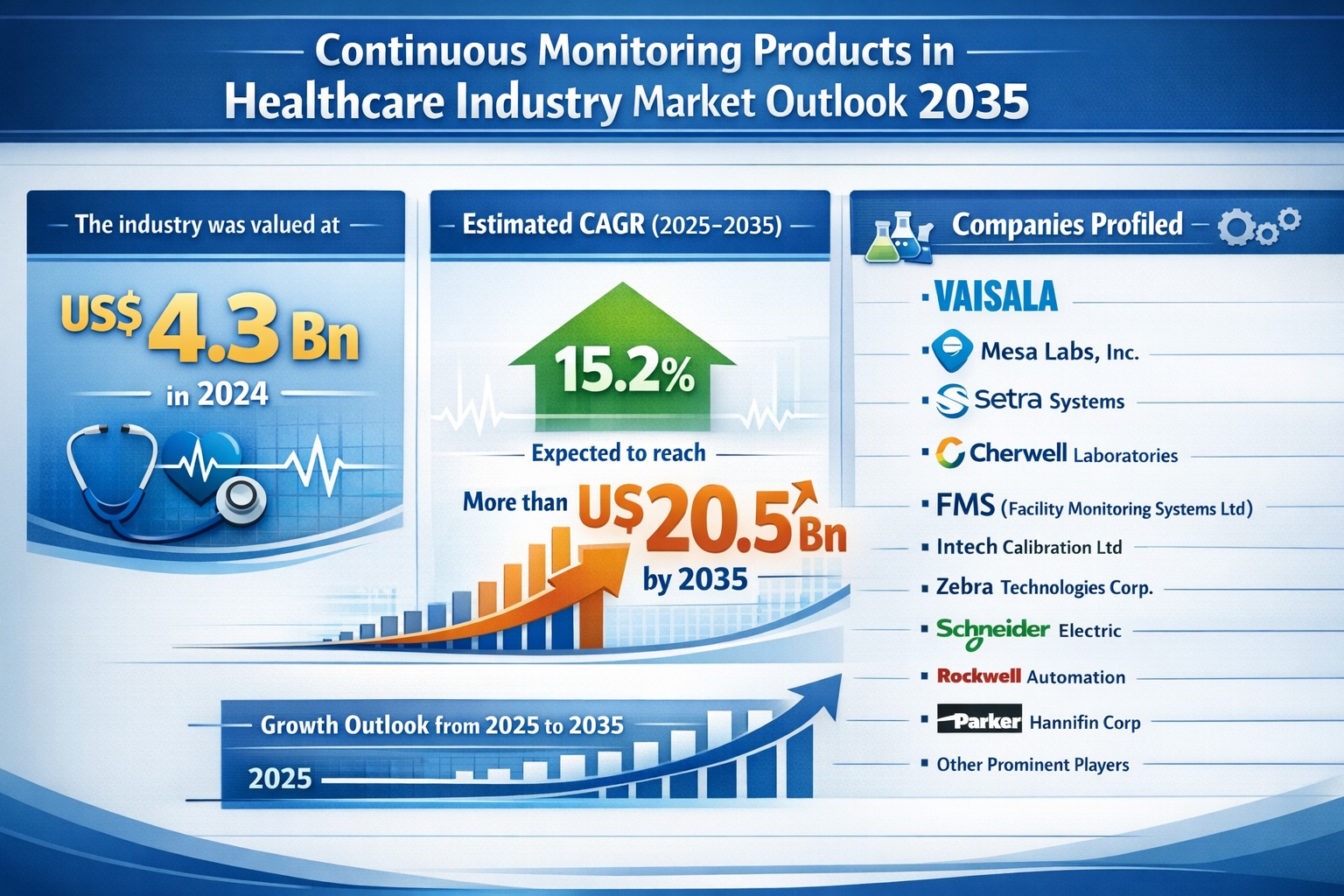
The global continuous monitoring products in healthcare industry market was valued at US$ 4.3 Bn in 2024 and is projected to grow at a remarkable compound annual growth rate (CAGR) of 15.2% from 2025 to 2035, reaching more than US$ 20.5 Bn by the end of 2035. The double-digit growth trajectory underscores the expanding adoption of real-time environmental and patient monitoring solutions across healthcare, biopharma manufacturing, and medical device production sectors.
Between 2020 and 2024, the market experienced steady growth driven by increasing digitization in healthcare infrastructure, growing awareness of healthcare-associated infections (HAIs), and the expansion of cold-chain logistics for temperature-sensitive pharmaceuticals such as vaccines and biologics. The forecast period from 2025 to 2035 is expected to witness accelerated adoption due to regulatory pressures, AI-enabled innovations, and the rapid deployment of cloud-based monitoring systems.
→Discover Market Opportunities – Request Your Sample Copy Now: https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=86609
→Analyst Viewpoint
Increasing adoption of remote monitoring for enhanced operational efficiency and regulatory compliance and quality assurance are some of the leading factors driving continuous monitoring products in healthcare industry market share. Continuous monitoring in healthcare leverages automated systems and smart technologies to track critical parameters such as temperature, humidity, air quality, and pressure in real time.
These solutions ensure pharmaceutical efficacy, contamination prevention, and improved patient care. In hospitals, they enable early detection of patient health deterioration, while in biopharma and medical device manufacturing, they help maintain GMP compliance and sterility standards.
→Market Overview
Continuous monitoring products in healthcare refer to the real-time monitoring and evaluation of patients’ vital signs, environmental conditions, and operational parameters using advanced sensors, wearable devices, and remote monitoring systems. These technologies collect and analyze data continuously, allowing healthcare professionals to identify abnormalities at an early stage.
Such systems are critical in intensive care units (ICUs), chronic disease management programs, post-surgical recovery units, laboratories, vaccine storage facilities, and sterile manufacturing environments. Monitoring devices track heart rate, blood pressure, oxygen saturation, respiratory rate, temperature, humidity, and particulate matter levels.
One of the most important advantages of continuous monitoring is enhanced patient safety and improved treatment outcomes. In ICUs, continuous monitoring devices instantly alert clinicians in the event of sudden fluctuations in vital signs. In home healthcare settings, patients with diabetes or cardiovascular diseases benefit from remote monitoring, reducing unnecessary hospital admissions and lowering healthcare costs.
In manufacturing settings, continuous environmental monitoring ensures that drugs, biologics, and sterile medical devices are produced under optimal conditions, safeguarding product quality and patient safety.
→Key Market Growth Drivers
1.Increasing Adoption of Remote Monitoring for Enhanced Operational Efficiency
Healthcare providers and manufacturers are increasingly deploying remote monitoring solutions to streamline operations and reduce manual intervention. Automated data logging eliminates human error and enhances workflow efficiency. Centralized dashboards allow organizations to monitor multiple facilities in real time, significantly improving response times and operational transparency.
2.Regulatory Compliance and Quality Assurance
Stringent regulatory frameworks imposed by authorities such as the FDA, EMA, WHO, and CDC are significantly influencing market expansion. Healthcare and pharmaceutical organizations must comply with Good Manufacturing Practices (GMP), Good Distribution Practices (GDP), and Good Laboratory Practices (GLP).
Environmental conditions must be maintained within strict parameters to prevent contamination or degradation. Continuous monitoring systems generate tamper-proof audit trails, ensuring organizations can demonstrate compliance during inspections. Failure to meet regulatory standards can lead to product recalls, reputational damage, and financial penalties, reinforcing the need for reliable monitoring solutions.
3.Growth in Biopharma and Medical Device Manufacturing
Rapid expansion of biologics production, vaccine manufacturing, and sterile medical device production is creating strong demand for temperature and humidity monitoring systems in cleanrooms and storage facilities.
4.Rising Healthcare-Associated Infections (HAIs)
Post-COVID-19, healthcare institutions are investing heavily in infection control infrastructure. Continuous monitoring systems help maintain sterile environments in ICUs, NICUs, surgical suites, and isolation rooms.
5.Continuous Monitoring Products in Healthcare Industry Market Revenue Growth
In line with the latest market trends, key players are investing in innovation and strategic partnerships. AI-driven predictive monitoring solutions, cloud-based data management platforms, and wearable health-monitoring devices are gaining traction.
For example, BioIntelliSense Inc. launched the BioButton Rechargeable in April 2024. This medical-grade wearable device monitors over 20 vital signs and provides up to 30 days of continuous monitoring on a single charge. Such innovations are significantly enhancing patient safety and remote monitoring capabilities.
→Market Segmentation with Regional Insights
By offerings, the equipment segment currently leads the market. This includes sensors, data loggers, environmental monitoring systems, and wearable devices. The demand for precise monitoring of temperature, humidity, pressure, and air quality is driving equipment sales across hospitals, laboratories, and pharmaceutical facilities.
The software segment is also witnessing strong growth. Cloud-integrated platforms provide centralized monitoring, automated alerts, predictive analytics, and remote access. The integration of IoT and AI in monitoring software enhances data accuracy and operational efficiency.
By setup, the market is divided into integrated systems and self-provisioning systems. Integrated systems are widely adopted by large hospitals and pharmaceutical manufacturers, while smaller clinics may opt for modular self-provisioning systems.
By end-use industry, the market serves biopharma manufacturing, medical device manufacturing, and hospitals and clinics. Biopharma manufacturing accounts for a substantial share due to strict GMP requirements.
Regionally, North America held the largest share in 2024. Strict regulatory enforcement by agencies such as FDA, CDC, CMS, and OSHA supports strong adoption. Europe follows closely, driven by EU MDR regulations and strong healthcare infrastructure. Asia Pacific is expected to witness the fastest growth between 2025 and 2035 due to expanding pharmaceutical manufacturing in China and India and rising healthcare investments. Latin America and Middle East & Africa are emerging markets with increasing adoption of digital health technologies.
→Market Challenges & Opportunities
1.Challenges
- High initial investment costs for advanced monitoring infrastructure
- Integration complexities with legacy hospital IT systems
- Data privacy and cybersecurity concerns
- Limited awareness in developing regions
2.Opportunities
- Expansion of telehealth and remote patient monitoring
- AI-powered predictive maintenance and analytics
- Growth in biologics and vaccine cold-chain logistics
- Increasing adoption of IoT-enabled smart hospitals
The integration of blockchain for secure data logging and AI for predictive alerts presents transformative growth opportunities over the next decade.
→Explore Strategies & Trends – Request Full Report Access – https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=86609
→Analysis of Key Players – Key Player Strategies
Prominent companies operating in the continuous monitoring products in healthcare industry market include Vaisala, Mesa Labs, Inc., Setra Systems, Cherwell Laboratories, FMS (Facility Monitoring Systems Ltd), Intech Calibration Ltd, Zebra Technologies Corp., Schneider Electric, Rockwell Automation, and Parker Hannifin Corp.
→Key Player Strategies
- Strategic partnerships with biopharma manufacturers and hospital networks
- Investment in AI-enabled predictive analytics
- Expansion of global distribution networks
- Development of cloud-based centralized monitoring platforms
- Focus on regulatory-compliant and audit-ready solutions
These players are enhancing their portfolios through mergers, product innovation, and geographic expansion to strengthen market presence.
→Recent Developments
In November 2024, Aranet expands its professional environmental monitoring lineup with the new Aranet 4xT-Probe Sensor, offering advanced, multi-point temperature monitoring solutions. This sensor enhances temperature monitoring precision with four external probes, allowing for simultaneous measurements across multiple locations. It is designed for applications requiring continuous temperature monitoring, such as HVAC systems for heating and ventilation.
In July 2024, Yardstik, a measurably better background screening company, has unveiled a new product called Continuous Monitoring that expands its compliance offering. It’s one more tool the company offers its customers to protect against bad actors within their organizations. Continuous Monitoring is a premium product for companies. Those who require more regular monitoring or are looking for a way to better manage reputation risks can leverage the 24/7 monitoring product instead of one-time background reports.
→Investment Landscape and ROI Outlook
The continuous monitoring products market presents strong ROI potential due to recurring revenue models from software subscriptions, maintenance services, and compliance audits. Investors are increasingly focusing on AI-driven healthtech startups and IoT-enabled monitoring solutions.
With a projected market expansion from US$ 4.3 Bn in 2024 to over US$ 20.5 Bn in 2035, the sector offers substantial long-term returns. Venture capital funding and strategic acquisitions are expected to rise significantly over the next decade.
→Why Buy This Report?
- Comprehensive analysis of market size and forecast from 2025 to 2035
- Detailed insights into drivers, restraints, opportunities, and trends
- In-depth regional and segment analysis
- Competitive landscape and company profiling
- Strategic recommendations for stakeholders
- Historical data from 2020 to 2024
- Electronic (PDF) + Excel format availability
This report enables healthcare providers, manufacturers, investors, and policymakers to make data-driven decisions and capitalize on emerging growth opportunities.
→FAQs
Q.How big was the global continuous monitoring products in healthcare industry market in 2024?
A.The market was valued at US$ 4.3 Bn in 2024.
Q.How is the market expected to grow during the forecast period?
A.It is projected to surpass US$ 20.5 Bn by the end of 2035, growing at a CAGR of 15.2% from 2025 to 2035.
Q.What are the key factors driving demand?
A.Increasing adoption of remote monitoring for enhanced operational efficiency and regulatory compliance and quality assurance.
Q.Which region holds the largest market share?
A.North America accounted for the largest share in 2024 and is expected to maintain dominance during the forecast period.
Q.Who are the prominent market players?
A.Leading companies include Vaisala, Mesa Labs, Inc., Setra Systems, Cherwell Laboratories, FMS (Facility Monitoring Systems Ltd), Intech Calibration Ltd, Zebra Technologies Corp., Schneider Electric, Rockwell Automation, Parker Hannifin Corp, and other prominent players.
Buy Full Report Now: https://www.transparencymarketresearch.com/checkout.php?rep_id=86609<ype=S
→Explore Latest Research Reports by Transparency Market Research:
Surgical Kits Market: https://www.transparencymarketresearch.com/surgical-kits-market.html
Surgical Tables and Lights Market: https://www.transparencymarketresearch.com/surgical-tables-lights-market.html
Viral Vector Production Market: https://www.transparencymarketresearch.com/viral-vector-production-market.html
About Transparency Market Research
Transparency Market Research, a global market research company registered at Wilmington, Delaware, United States, provides custom research and consulting services. Our exclusive blend of quantitative forecasting and trends analysis provides forward-looking insights for thousands of decision makers. Our experienced team of Analysts, Researchers, and Consultants use proprietary data sources and various tools & techniques to gather and analyses information.
Our data repository is continuously updated and revised by a team of research experts, so that it always reflects the latest trends and information. With a broad research and analysis capability, Transparency Market Research employs rigorous primary and secondary research techniques in developing distinctive data sets and research material for business reports.



Leave a Reply