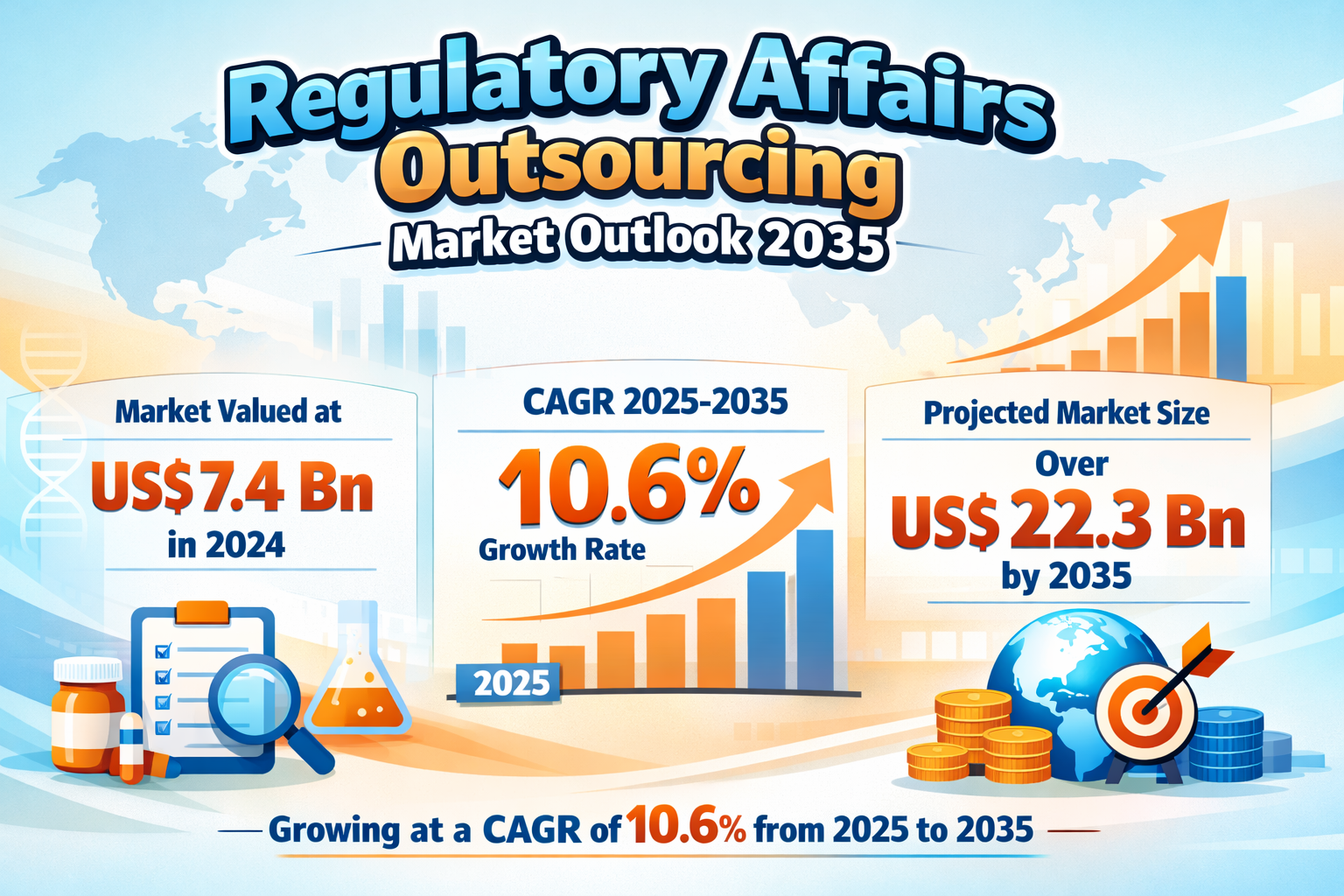
The global Regulatory Affairs Outsourcing Market was valued at US$ 7.4 Billion in 2024 and is projected to grow at a compound annual growth rate (CAGR) of 10.6% from 2025 to 2035, reaching more than US$ 22.3 Billion by the end of 2035. The robust expansion reflects the increasing complexity of global regulatory frameworks, the growing volume of clinical trials, and the strategic shift among pharmaceutical, biotechnology, and medical device companies toward outsourcing regulatory functions.
Unlock key takeaways and conclusions from our Report in this sample –
https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=3528
As regulatory pathways become more data-intensive and geographically nuanced, organizations are increasingly relying on specialized external partners to ensure timely product approvals and compliance with evolving international standards.
Market Overview
Regulatory affairs outsourcing involves collaborating with third-party specialists to manage regulatory submissions, compliance processes, product registrations, and lifecycle documentation across various global markets. These services support pharmaceutical, biopharma, biotech, and medical device companies in navigating the intricate web of global regulations governing product development, clinical testing, approval, and commercialization.
The outsourcing model provides access to experienced regulatory consultants, localized legal representation, clinical trial application experts, and submission specialists. It enables organizations to reduce operational overheads, mitigate compliance risks, accelerate time-to-market, and optimize internal resources.
The market is segmented by:
- Service Type
- Product Development Stage
- Enterprise Size
- Therapeutic Area
- End-user
- Region
North America currently dominates the global landscape with a market share of 39.4% in 2024, while Asia Pacific is expected to emerge as the fastest-growing regional market during the forecast period.
Key Market Growth Drivers
- Increasing Stringent Government Regulations
Rapid advancements in biotechnology, pharmaceuticals, and medical devices have prompted governments worldwide to tighten regulatory frameworks. Regulatory authorities now require more extensive safety, efficacy, and quality data prior to product approvals. This has significantly increased the complexity of regulatory submissions.
For instance, in November 2024, the Medical Device Coordination Group (MDCG) issued Q&A addressing practical aspects of the gradual roll-out of Eudamed under Regulations (EU) 2017/745 (MDR) and (EU) 2017/746 (IVDR), as amended by Regulation (EU) 2024/1860. The Q&A specify that Eudamed includes six modules and clarify that the obligations and requirements for each module will become applicable six months after the publication of the notice confirming the functionality of the module in the Official Journal.
Such regulatory updates demand constant monitoring and proactive compliance strategies, encouraging companies to outsource regulatory tasks to specialized firms.
- Demand for Local Representation in High-Growth Markets
Global expansion strategies require companies to comply with region-specific regulatory requirements. Emerging markets across Asia Pacific, Latin America, and the Middle East have distinct approval pathways and documentation standards.
Outsourcing firms provide localized expertise, regional offices, and direct liaison capabilities with regulatory authorities. This is especially crucial in markets like China, India, and Japan, where regulatory reforms are evolving rapidly.
For example, in March 2025, ICON PLC expanded its outsourcing services within the Asia Pacific region. This strategic move responds directly to the growing demand for regulatory consultation and clinical trial support in rapidly developing markets such as China and India.
- Growth in Clinical Trials and Globalization of Medical Devices
The Product Registration and Clinical Trial Applications segment dominates the service type category. Increasing global clinical trial activity, along with regulatory reforms in emerging markets, is driving demand for outsourced regulatory submission and legal representation services.
Analysis of Key Players – Key Player Strategies
The competitive landscape is characterized by global contract research organizations (CROs), regulatory consultancies, and specialized compliance firms. Prominent companies include:
- Accell Clinical Research, LLC
- Genpact
- CRITERIUM, INC
- Promedica International
- WuXi AppTec
- Medpace
- Charles River Laboratories
- ICON plc
- Labcorp Drug Development
- Parexel International Corporation
- Freyr
- PHARMALEX GMBH
- Other Prominent Players
Strategic Focus Areas
- End-to-End Service Expansion: Leading players are broadening portfolios to include clinical trial support, pharmacovigilance, compliance management, and lifecycle services.
- Regional Expansion: Companies are strengthening footprints in Asia Pacific and Latin America.
- Digital Transformation: Investment in regulatory intelligence platforms and submission automation tools.
- Mergers & Acquisitions: Acquisition of niche consultancies to enhance therapeutic or geographic capabilities.
- Strategic Partnerships: Collaborations with hospitals, research institutes, and specialty clinics.
Market Challenges & Opportunities
Challenges
- Regulatory Variability Across Regions
Differing approval timelines and documentation standards complicate global submissions. - Data Privacy and Compliance Risks
Handling sensitive clinical data across jurisdictions requires stringent cybersecurity protocols. - High Dependency on Skilled Workforce
The market relies heavily on experienced regulatory professionals, creating talent competition.
Opportunities
- Emerging Market Expansion
Rapid healthcare infrastructure investments in Asia Pacific and Latin America present growth avenues. - Lifecycle Management Services
Increasing focus on post-market surveillance and compliance monitoring opens new revenue streams. - Digital Regulatory Solutions
Automation, AI-based submission tools, and centralized documentation systems enhance efficiency.
Recent Developments (As per Report)
In August 2024, LEAP Consulting Group, a boutique digital consultancy servicing the clinical laboratory industry, is excited to announce its offering for clinical laboratories already established as CLIA certified to facilitate the planning and remediation required to extend their existing CLIA, CAP, and NYSDOH CLEP to meet the expanded requirements as recently announced by the U.S. Food and Drug Administration (FDA) as part of the FDA’s LDT Final Rule announcement declaration in May 2024.
In October 2024, ProductLife Group (PLG), a global specialist in regulatory, scientific, compliance, and digital transformation consulting services for the life sciences industry, announced the acquisition of Callisto, a UK-based consultancy specializing in Regulatory Affairs (RA), Pharmacovigilance (PV) and GMDP services within multiple regulated sectors including human and veterinary medicines, medical devices, and borderline products.
Investment Landscape and ROI Outlook
The regulatory affairs outsourcing market presents a high-growth investment opportunity driven by:
- Double-digit CAGR (10.6%)
- Increasing regulatory complexity
- Global expansion of pharmaceutical manufacturing
- Rising R&D expenditure
Investments in digital regulatory platforms and emerging markets are expected to generate strong ROI due to scalable service models and recurring compliance needs.
Private equity firms and strategic investors are increasingly targeting mid-sized regulatory consultancies for acquisitions to consolidate market share and expand geographic reach.
Market Segmentation
By Service Type
- Regulatory Consulting and Legal Representation
- Product Registration and Clinical Trial Applications (Dominant)
- Regulatory Writing and Publishing
- Regulatory Submission
- Regulatory Operations
- Others (Post Approval/Lifecycle Management)
By Product Development Stage
- Preclinical
- Clinical
- PMA (Post Market Authorization)
By Enterprise Size
- Small/Medium Enterprises
- Large Enterprises
By Therapeutic Area
- Oncology
- Neurology
- Cardiology
- Immunology
- Dermatology
- Others (Infectious disorders, etc.)
By End-user
- Medical Device Companies
- Biopharma & Pharmaceutical Companies
- Others (Biotech Companies)
By Region
- North America (Leading Region – 39.4% Share in 2024)
- Europe
- Asia Pacific (Fastest Growing)
- Latin America
- Middle East & Africa
Why Buy This Report?
- Comprehensive 2020–2035 Market Analysis
- Detailed Segment & Regional Insights
- Competitive Landscape & Company Profiles
- Growth Drivers, Challenges & Opportunities
- Investment and Strategic Planning Insights
- Excel + PDF Format for Data Analysis
- Actionable Intelligence for Decision Makers
This report provides an in-depth understanding of the regulatory outsourcing ecosystem, enabling stakeholders to make informed strategic decisions.
Frequently Asked Questions (FAQs)
- What is driving growth in the Regulatory Affairs Outsourcing Market?
Increasing stringent government regulations, growing clinical trial activity, and demand for localized regulatory representation are the primary growth drivers. - Which region leads the global market?
North America leads the market with a 39.4% share in 2024, while Asia Pacific is the fastest-growing region. - Which service segment dominates the market?
Product Registration and Clinical Trial Applications dominate due to rising clinical trial volumes and global product approvals. - Who are the key players in the market?
Major players include ICON plc, WuXi AppTec, Charles River Laboratories, Parexel International Corporation, Genpact, and others. - What is the forecast market value by 2035?
The market is projected to exceed US$ 22.3 Billion by the end of 2035.



Leave a Reply