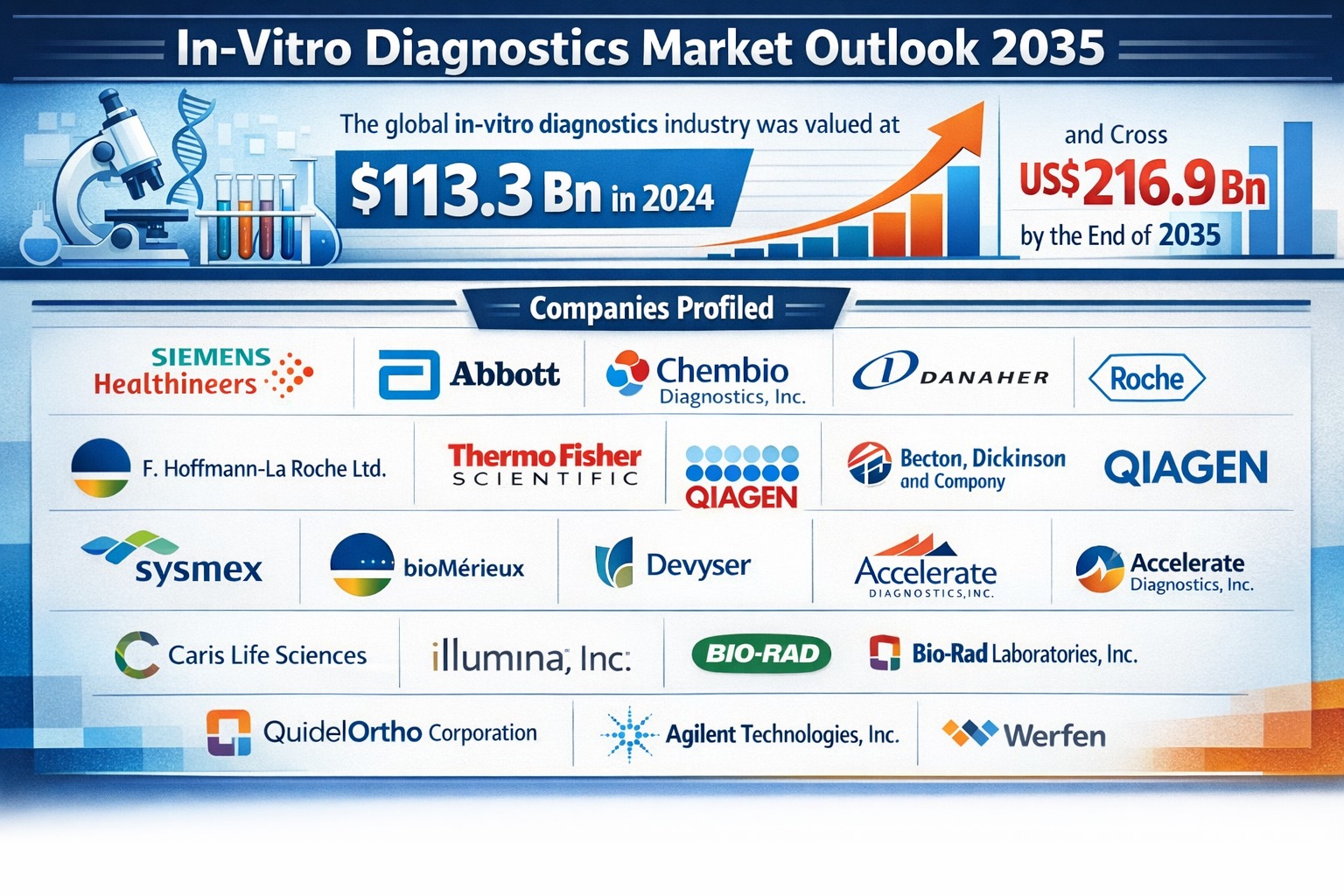
The global in-vitro diagnostics (IVD) market continues to demonstrate strong growth momentum as healthcare systems worldwide increasingly prioritize early disease detection, personalized medicine, and rapid diagnostic solutions. The market was valued at US$ 113.3 billion in 2024 and is projected to expand at a compound annual growth rate (CAGR) of 6.1% from 2025 to 2035. By the end of the forecast period, the global IVD industry is expected to surpass US$ 216.9 billion by 2035.
This expansion reflects the growing demand for accurate diagnostic testing in hospitals, laboratories, and home care environments. As healthcare systems shift toward preventive medicine and value-based healthcare models, diagnostic technologies are becoming indispensable for clinical decision-making and patient management.
The steady growth trajectory of the industry is also supported by technological innovations in molecular diagnostics, automation, artificial intelligence-assisted analysis, and digital health integration, which are transforming the speed and accuracy of diagnostic processes.
→Discover Market Opportunities – Request Your Sample Copy Now: https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=12959
Market Overview
In vitro diagnostics are medical tests performed on biological samples such as blood, urine, saliva, or tissue collected from the human body. These tests play a critical role in identifying diseases, monitoring health conditions, and guiding treatment decisions.
IVD testing encompasses a wide array of diagnostic technologies, including molecular diagnostics, clinical chemistry, immunoassays, hematology testing, microbiology diagnostics, and urinalysis. These technologies enable healthcare providers to detect diseases at early stages and monitor therapeutic responses more effectively.
The importance of IVD solutions has grown significantly in recent years due to the increasing global burden of chronic diseases and emerging infectious diseases. In addition, the integration of genomics and precision medicine has accelerated the adoption of advanced diagnostic tools that can identify genetic variations and guide targeted therapies.
Point-of-care testing (POCT) and home-based diagnostics are also reshaping the healthcare landscape by enabling faster testing and improving accessibility for patients. With ongoing innovations in miniaturization, automation, and digital connectivity, IVD systems are becoming more efficient and accessible, expanding their role in global healthcare systems.
Analysts’ Viewpoint
The global in vitro diagnostics market is witnessing steady growth driven by increasing prevalence of chronic and infectious diseases, growing awareness regarding early and accurate disease detection, and rapid advancements in diagnostic technologies such as molecular diagnostics, next-generation sequencing (NGS), and point-of-care testing.
The transition toward personalized medicine and precision diagnostics is further accelerating the adoption of IVD technologies across multiple therapeutic areas including oncology, cardiology, and infectious diseases. Additionally, expanding healthcare infrastructure in developing economies and increased government investments in healthcare diagnostics are expected to create new growth opportunities.
Technological convergence involving artificial intelligence, digital health platforms, and home-based testing solutions is transforming the diagnostic ecosystem by making testing faster, more accurate, and more patient-centric.
However, challenges such as high development costs of advanced diagnostic tools and complex regulatory frameworks in different regions may slow market penetration. Despite these barriers, the long-term outlook of the industry remains highly positive due to continuous technological innovation and increasing clinical demand worldwide.
Key Market Growth Drivers
Rising Prevalence of Chronic and Infectious Diseases
The increasing incidence of chronic diseases such as diabetes, cardiovascular disorders, and cancer is one of the most significant drivers of the IVD market. Aging populations, sedentary lifestyles, and unhealthy dietary habits have contributed to the growing global burden of chronic illnesses.
According to data published by the Centers for Disease Control and Prevention (CDC) in October 2024, approximately 6 in 10 Americans suffer from at least one chronic disease, while 4 in 10 individuals live with two or more chronic conditions.
In addition, infectious diseases continue to pose major global health challenges. Emerging viral infections and re-emerging diseases such as tuberculosis, hepatitis, and sexually transmitted infections require rapid and accurate diagnostic tools for effective disease management.
The CDC reported that over 2.5 million cases of gonorrhea, chlamydia, and syphilis were recorded in the United States in 2022, highlighting the increasing need for advanced diagnostic technologies.
Growing Demand for Early, Accurate, and Non-Invasive Diagnostic Tools
Healthcare providers and patients increasingly prefer diagnostic tests that deliver quick, reliable, and minimally invasive results. Technological innovations have enabled the development of diagnostic methods that require only small volumes of blood or saliva samples while providing highly accurate outcomes.
These tests allow clinicians to make timely treatment decisions and improve patient outcomes. Rapid diagnostic tools also reduce hospitalization time and healthcare costs, thereby increasing their adoption in hospitals, clinics, and home testing environments.
Technological Advancements in Molecular Diagnostics and POCT
Advances in molecular diagnostics, digital PCR, and next-generation sequencing have significantly enhanced diagnostic accuracy and efficiency. These technologies enable precise detection of genetic mutations, infectious pathogens, and biomarkers associated with various diseases.
Point-of-care testing devices are also gaining popularity because they provide immediate diagnostic results at the patient’s bedside or in remote healthcare settings. The combination of these technologies with artificial intelligence and digital data management platforms is further enhancing diagnostic capabilities.
Reagents & Kits Segment Driving Market Growth
Among product categories, reagents and kits represent a significant share of the in-vitro diagnostics market. These consumables are essential components used in diagnostic testing procedures.
Reagents typically include enzymes, antibodies, buffers, and chemical solutions that react with patient samples to produce measurable responses. Diagnostic kits combine these reagents with instructions and accessories required for specific tests.
The increasing volume of diagnostic testing worldwide has significantly boosted demand for reagents and kits. As instruments are installed in hospitals and laboratories, the demand for consumables continues to grow because they must be replenished regularly.
Furthermore, advancements in reagent formulation and kit design are enabling faster, more sensitive, and cost-effective diagnostic testing, further strengthening this segment’s growth.
→Explore Strategies & Trends – Request Full Report Access – https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=12959
Regional Outlook
North America currently leads the global in-vitro diagnostics market and is expected to maintain its dominant position throughout the forecast period.
The region benefits from advanced healthcare infrastructure, strong research and development capabilities, and rapid adoption of next-generation diagnostic technologies. The United States plays a particularly important role in driving market growth due to the presence of leading diagnostic companies and extensive healthcare funding.
Regulatory oversight by the U.S. Food and Drug Administration (FDA) ensures high standards for diagnostic products while also supporting innovation in medical technologies.
Europe represents another significant market for IVD solutions, supported by strong healthcare systems and increasing adoption of molecular diagnostics. Meanwhile, Asia Pacific is emerging as a rapidly growing market due to expanding healthcare infrastructure, increasing population, and rising demand for diagnostic testing in countries such as China and India.
Latin America and the Middle East & Africa are also expected to witness gradual growth as healthcare accessibility improves and governments invest in diagnostic capabilities.
Analysis of Key Players – Key Player Strategies
The competitive landscape of the global in-vitro diagnostics market includes a large number of multinational corporations and emerging biotechnology companies. Major players such as Siemens Healthineers AG, Abbott, Danaher Corporation, F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific, Becton Dickinson and Company, QIAGEN N.V., Sysmex Corporation, bioMérieux, Illumina, Bio-Rad Laboratories, Agilent Technologies, and Werfen play a critical role in shaping industry dynamics.
These companies focus heavily on research and development, strategic partnerships, and product innovation to maintain their competitive edge. Investments in molecular diagnostics, next-generation sequencing technologies, and digital diagnostic platforms are among the primary strategies used by market leaders.
Additionally, many companies are expanding their global presence through acquisitions and collaborations with biotechnology firms and pharmaceutical companies.
Market Challenges & Opportunities
Despite strong growth prospects, the in-vitro diagnostics industry faces several challenges. One of the primary barriers is the high development and implementation cost of advanced diagnostic technologies, which can limit adoption in low-income regions.
Regulatory complexity is another challenge. Strict regulatory requirements such as the European Union’s In Vitro Diagnostic Regulation (IVDR) and evolving FDA regulations for laboratory-developed tests can delay product approvals and market entry.
Data privacy concerns related to digital diagnostic platforms and patient health data also present challenges for industry players.
However, these challenges also create opportunities for innovation. The development of decentralized diagnostic solutions, home-based testing systems, and digital diagnostic platforms presents significant growth potential.
Local manufacturing of reagents in emerging markets, particularly in Asia, and collaborations between diagnostic companies and digital health providers are expected to drive future market expansion.
Key Player Strategies
Key companies in the IVD market employ several strategic initiatives to strengthen their market position. These include expanding product portfolios, launching innovative diagnostic technologies, and investing in advanced molecular diagnostics platforms.
Partnerships with pharmaceutical companies for companion diagnostics development are also becoming increasingly common, especially in the field of oncology and personalized medicine.
Companies are also focusing on expanding their presence in emerging markets through localized manufacturing and distribution networks to meet the growing demand for diagnostic solutions.
Recent Developments
In September 2024, QIAGEN introduced the QIAcuityDx Digital PCR System, a digital PCR solution designed for clinical diagnostics. The system and related products are FDA-exempt for use in the United States and IVDR-approved for the European market. The system provides highly accurate quantitation of target DNA and RNA and is particularly useful for monitoring cancer development. QIAGEN is expanding its application menu and collaborating with pharmaceutical companies for companion diagnostics.
In January 2024, ELITechGroup, a Bruker Company, launched the GI Bacterial PLUS ELITe MGB Kit. The assay is designed to diagnose gastrointestinal bacterial infections and targets pathogens such as Campylobacter spp., Clostridium difficile, Salmonella spp., Shigella spp., and Yersinia enterocolitica.
Investment Landscape and ROI Outlook
The investment landscape in the in-vitro diagnostics industry remains highly attractive due to the sector’s strong growth prospects and critical role in modern healthcare.
Investments are increasingly directed toward molecular diagnostics, digital diagnostic platforms, artificial intelligence-enabled testing, and point-of-care devices. Venture capital firms, pharmaceutical companies, and healthcare investors are actively funding startups and research initiatives focused on innovative diagnostic technologies.
The expected market growth from US$ 113.3 billion in 2024 to over US$ 216.9 billion by 2035 indicates strong return-on-investment potential for stakeholders. Companies that successfully integrate digital technologies, automation, and precision diagnostics into their product offerings are expected to achieve higher profitability and market share.
Market Segmentations
The in-vitro diagnostics market can be segmented based on product and services, technology, application, end-user, and region.
By product and services, the market includes instruments, reagents and kits, services, and data management software.
By technology, key segments include immunoassay, clinical chemistry, molecular diagnostics, hematology, coagulation and hemostasis testing, microbiology diagnostics, urinalysis, and other technologies.
By application, the market covers oncology, infectious diseases, diabetes, cardiology, nephrology, drug testing and pharmacogenomics, autoimmune diseases, and other diagnostic areas.
By end user, the industry serves hospital laboratories, clinical laboratories, point-of-care testing centers, academic institutes, home care settings, and other healthcare facilities.
Regionally, the market spans North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa, with North America currently leading the global industry.
Why Buy This Report?
This report provides comprehensive insights into the global in-vitro diagnostics market, including detailed analysis of industry trends, market drivers, and competitive dynamics.
It offers in-depth information on market size, growth forecasts, and emerging technologies shaping the diagnostic industry. The report also provides extensive analysis of leading companies, their strategic initiatives, and recent developments.
Additionally, the report includes regional insights, segmentation analysis, and investment opportunities, making it a valuable resource for investors, healthcare organizations, diagnostic manufacturers, and policymakers.
FAQs
- How big was the global In-Vitro diagnostics market in 2024?
The global in-vitro diagnostics market was valued at US$ 113.3 billion in 2024. - What is the projected size of the In-Vitro diagnostics market by 2035?
The market is expected to exceed US$ 216.9 billion by the end of 2035. - What factors are driving the growth of the In-Vitro diagnostics market?
Key growth drivers include the rising prevalence of chronic and infectious diseases, increasing demand for early and non-invasive diagnostic tools, and technological advancements in molecular diagnostics and point-of-care testing. - What is the expected CAGR of the In-Vitro diagnostics market during the forecast period?
The market is anticipated to grow at a CAGR of 6.1% from 2025 to 2035. - Which region dominates the global In-Vitro diagnostics market?
North America is expected to dominate the market due to its advanced healthcare infrastructure, strong research capabilities, and widespread adoption of diagnostic technologies.
Buy Full Report Now: https://www.transparencymarketresearch.com/checkout.php?rep_id=12959<ype=S
→Explore Latest Research Reports by Transparency Market Research:
Mechanical Ventilators Market: https://www.transparencymarketresearch.com/mechanical-ventilators-market.html
Skincare Devices Market: https://www.transparencymarketresearch.com/skincare-devices-market.html
Diagnostic Catheters Market: https://www.transparencymarketresearch.com/diagnostic-catheter-market.html
About Transparency Market Research
Transparency Market Research, a global market research company registered at Wilmington, Delaware, United States, provides custom research and consulting services. Our exclusive blend of quantitative forecasting and trends analysis provides forward-looking insights for thousands of decision makers. Our experienced team of Analysts, Researchers, and Consultants use proprietary data sources and various tools & techniques to gather and analyses information.
Our data repository is continuously updated and revised by a team of research experts, so that it always reflects the latest trends and information. With a broad research and analysis capability, Transparency Market Research employs rigorous primary and secondary research techniques in developing distinctive data sets and research material for business reports.


Leave a Reply