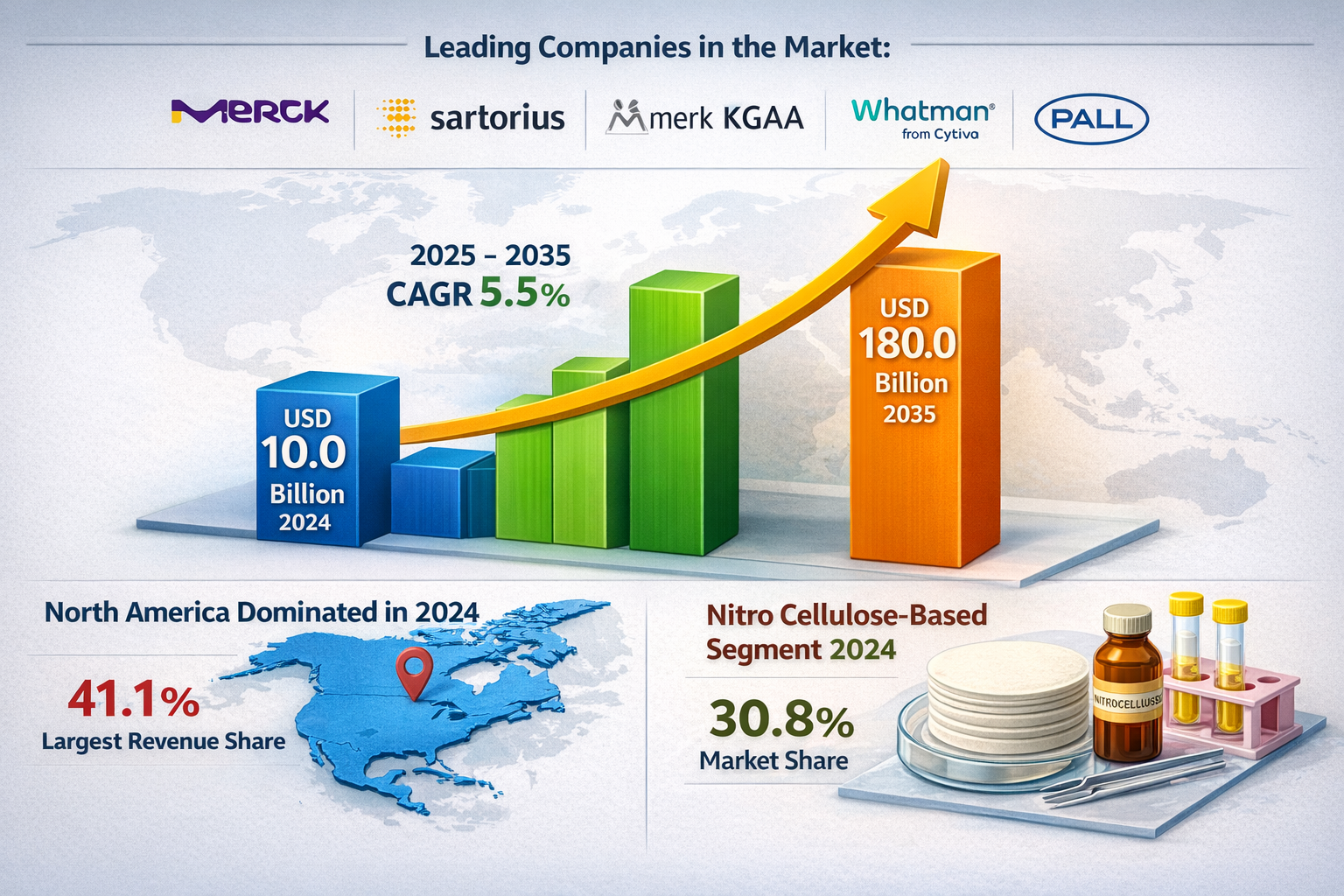
The global lateral flow assay market was valued at USD 10.0 billion in 2024 and is projected to reach USD 18.0 billion by 2035, growing at a CAGR of 5.5% during the forecast period from 2025 to 2035. The market growth is driven by increasing demand for rapid diagnostic testing, rising prevalence of infectious diseases, and expanding applications in point-of-care testing across healthcare settings.
👉 Get sample market research report copy today@ https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=39356
Market Overview
The lateral flow assay (LFA) market is largely driven by the demand for easy-to-use diagnostics that can produce rapid results without the need for a central lab infrastructure. The primary catalysts for growth include increased infectious disease testing programs, greater consumer acceptance of home-testing, and the desire to decentralize screening in low-resource environments. In response to increased demand, the manufacturing processes have been scaled, and the quality assurance has been improved, resulting in lower per-test costs and enhanced economic viability of LFAs for screening and surveillance.
The increased use case applications beyond infectious disease, as well as the basic biology of cardiac markers, fertility tests, drug residue screening and some oncology biomarkers, are increasing addressable markets and cross-industry adoption. Furthermore, funding from public and private sector partners for epidemic preparedness and procurement structures favoring rapid technologies common in procurement are creating recurring demand streams. Collectively, these factors have shifted LFAs from episodic, disease outbreak-based usage to implementing LFAs in more established roles in testing pathways of care in primary care and community health programs.
Analysts’ Viewpoint on the Global LFA Market
The global market for lateral-flow-assay (LFA) is a response to the increasing demand for point-of-care diagnostics, materials innovation, and decentralized delivery of healthcare. LFA components – mainly membranes, sample/conjugate/absorbent pads, labels, backing cards, and reagents – are the main determinants of the sensitivity, reproducibility, and cost of LFAs as a product. Therefore, the quality and reliability of component supply, specifically led by the diagnostic original-equipment manufacturers (OEMs) and contract-development manufacturers (CDMOs), will guide purchasers’ decisions.
Major growth factors are: adoption of infectious-disease testing; scaling routine screening programmes; increasing point-of-care testing in primary care and pharmacies; and demand for multiplex and quantitative LFAs, which will require next-level membranes and advanced labels. Major manufacturers of membranes, nanoparticles/labels, and contract manufacturing have good materials supply chains, influence market dynamics via product innovation, scale of economy, and technical best practices – e.g. membranes with more control of flow (more consistent results) or conjugate pads that improve stabilization reduce variability of test results, and the number of components phase development programs for kit manufacturers.
Analysis of Key Players in the Lateral Flow Assay Market
The LFA landscape is highly competitive, featuring major diagnostic conglomerates and specialized biotechnology firms.
Leading companies operating in the global market include:
- Merck KGaA
- Sartorius AG
- MilliporeSigma
- Cobetter Filtration Equipment Co., Ltd.
- Ahlstrom Corporation
- Advanced Microdevices Pvt. Ltd. (India)
- Axiva Sichem Pvt. Ltd. (India)
- DCN Dx
- Fortis Life Sciences LLC
- BioAssay Works LLC
- ProGnosis Biotech Pvt. Ltd.
- Cytodiagnostics Inc.
- NanoComposix
- Whatman (Cytiva)
- Pall Corporation
- Other Prominent Players
Strategic movements in 2026 are focused on supply chain resilience, with companies diversifying their sources for “White Gold” (platinum) and specialized membranes to avoid the volatility seen in early 2026.
Recent Developments in the Lateral Flow Assay Market
- In January 2025, BIOMÉRIEUX announced an agreement to acquire SpinChip Diagnostics ASA, a Norwegian diagnostics company known for its innovative immunoassay platform. This acquisition aims to bolster bioMérieux’s point-of-care diagnostics capabilities.
- In February 2024, BIOMÉRIEUX entered into a strategic research collaboration with the U.S. Food and Drug Administration (FDA) to develop advanced tools for detecting food-borne pathogens, including Shiga-toxin producing E. coli, Cyclospora cayetanensis, Salmonella spp., and Listeria monocytogenes.
👉 Discuss Implications for Your Industry Request Sample Research Report PDF@ https://www.transparencymarketresearch.com/sample/sample.php?flag=B&rep_id=39356
Key Market Trends & Strategies
📱 Smartphone & Digital Integration
- App-Based Testing: Patients can now scan their LFA strips using proprietary apps that provide a clear “Positive/Negative” result and automatically notify their physician.
- Cloud Connectivity: Real-time surveillance maps of infectious disease outbreaks (like seasonal flu) are being powered by anonymous data from connected LFA readers.
📈 Market Challenges
- Accuracy Perceptions: Despite improvements, LFAs still struggle with a “low accuracy” stigma compared to molecular (PCR) testing, particularly in early-stage infections.
- Regulatory Hurdles: Stricter FDA and EU MDR requirements for home-use diagnostic devices are increasing the R&D costs and time-to-market for new entrants.
🌟 Growth Opportunities
- Oncology & Cardiac Markers: Development of LFA-based “Liquid Biopsies” to detect early-stage cancer markers (like PSA or CEA) at home.
- Animal Health: The Veterinary LFA market is surging due to the rising “humanization of pets,” with owners demanding rapid diagnostic answers for pet allergies and infections.
Market Segmentation
By Product
- Kits & Reagents (~66% Share)
- Lateral Flow Readers (Digital, Handheld)
- LFA Components (Membranes, Pads)
By Application
- Clinical Testing (Infectious, Cardiac, Pregnancy)
- Veterinary Diagnostics
- Food Safety & Environment
By End-User
- Hospitals & Clinics
- Home Care Settings (Fastest Growth)
- Diagnostic Laboratories
Regional Outlook
- North America: The largest market (~37% share). Driven by a high volume of POC testing, favorable reimbursement policies for digital diagnostics, and the presence of industry giants like Abbott and Quidel.
- Asia-Pacific: The fastest-growing region. Expansion is fueled by large-scale government screening programs for malaria and tuberculosis, alongside a booming “local manufacturing” movement in China and India.
- Europe: Focuses on Antimicrobial Stewardship. LFAs are being heavily utilized in primary care to distinguish between viral and bacterial infections, reducing the unnecessary prescription of antibiotics.
Conclusion
The Lateral Flow Assay market in 2026 is moving far beyond its traditional roots. By merging biochemical simplicity with digital sophistication (AI and IoT), LFAs are becoming the frontline of a decentralized healthcare system. The future of the market lies in quantitative multiplexing—the ability to give a patient detailed, digital insights into multiple health markers from a single drop of blood or saliva, right at the kitchen table.
👉 To buy this comprehensive market research report, click here to inquire@ https://www.transparencymarketresearch.com/checkout.php?rep_id=39356<ype=S
FAQs
- How big was the global lateral flow assay market in 2024?
The global lateral flow assay market was valued at US$ 10.0 Bn in 2024
- How big will the global lateral flow assay industry be in 2035?
The global lateral flow assay industry is projected to reach more than US$ 18.0 Bn by the end of 2035
- What are the factors driving the lateral flow assay market?
Increasing demand for rapid infectious disease screening and technological advancements improving sensitivity and multiplexing
- What will be the CAGR of the global lateral flow assay industry during the forecast period?
The CAGR is anticipated to be 5.5% from 2025 to 2035
About Transparency Market Research
Transparency Market Research, a global market research company registered at Wilmington, Delaware, United States, provides custom research and consulting services. Our exclusive blend of quantitative forecasting and trends analysis provides forward-looking insights for thousands of decision makers. Our experienced team of Analysts, Researchers, and Consultants use proprietary data sources and various tools & techniques to gather and analyses information.
Our data repository is continuously updated and revised by a team of research experts, so that it always reflects the latest trends and information. With a broad research and analysis capability, Transparency Market Research employs rigorous primary and secondary research techniques in developing distinctive data sets and research material for business reports.
Contact:
Transparency Market Research Inc.
CORPORATE HEADQUARTER DOWNTOWN,
1000 N. West Street,
Suite 1200, Wilmington, Delaware 19801 USA
Tel: +1-518-618-1030
USA – Canada Toll Free: 866-552-3453
Website: https://www.transparencymarketresearch.com
Email: sales@transparencymarketresearch.com


Leave a Reply