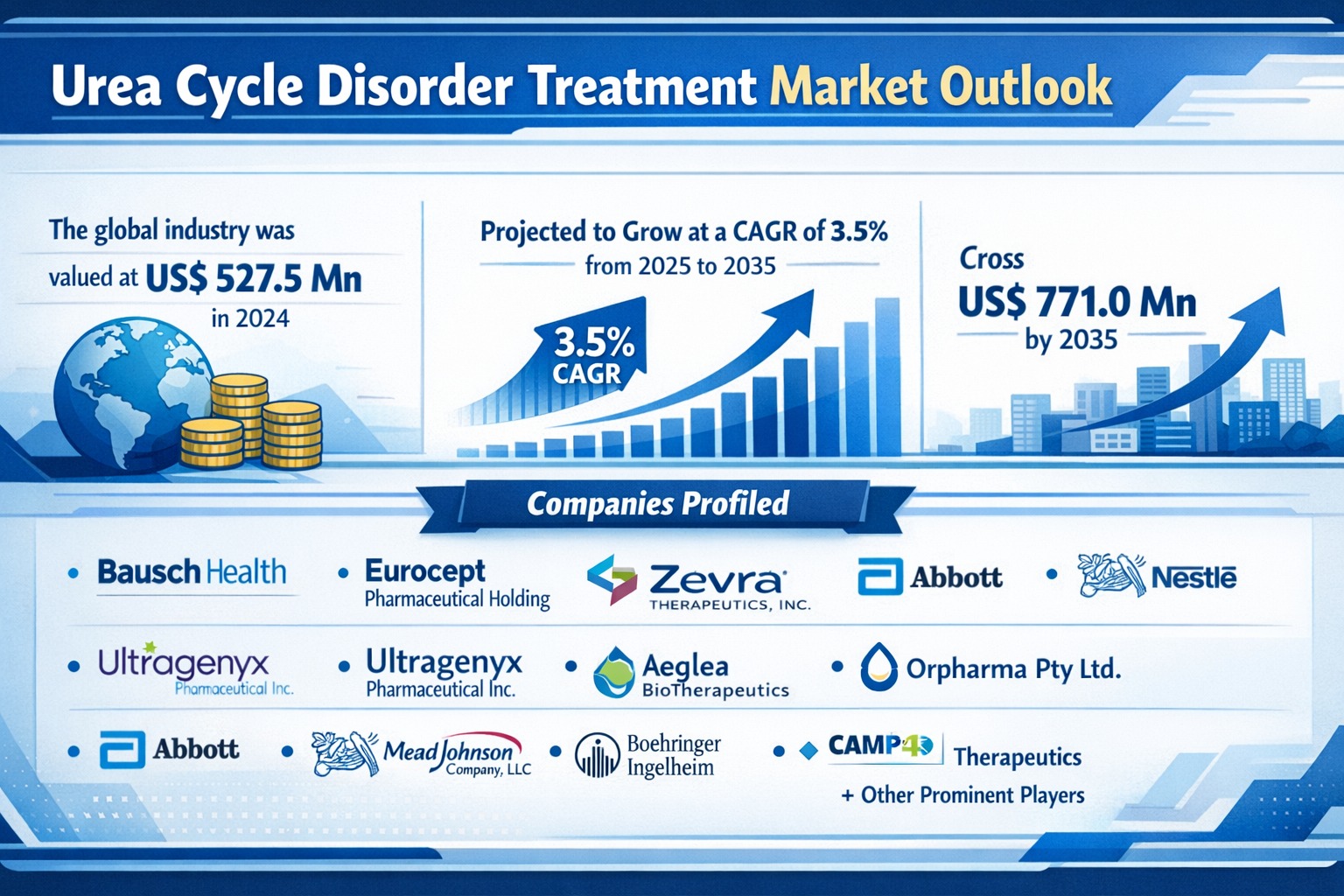
The global urea cycle disorder (UCD) treatment market was valued at US$ 527.5 Mn in 2024 and is projected to expand at a moderate CAGR of 3.5% from 2025 to 2035, surpassing US$ 771.0 Mn by the end of 2035. The steady expansion of the market reflects sustained demand for ammonia-lowering therapies, increasing diagnosis rates of rare metabolic disorders, and the gradual introduction of innovative treatment modalities including gene therapies and enzyme replacement drugs.
Between 2020 and 2024, the market demonstrated stable growth supported by expanding newborn screening programs and improved access to specialized metabolic clinics. Over the forecast period, incremental value addition of more than US$ 240 Mn is anticipated, underlining the long-term therapeutic need and growing research focus in rare genetic disorders.
→Discover Market Opportunities – Request Your Sample Copy Now: https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=86533
→Analysts’ Viewpoint Regarding Urea Cycle Disorder Treatment Market Scenario
The global urea cycle disorder treatment market is poised for consistent growth, driven by increasing prevalence of UCDs and a surge in product approvals. Although UCDs are rare genetic conditions, improved diagnostic technologies and enhanced awareness among healthcare professionals are leading to higher reported incidence rates. As more individuals receive confirmed diagnoses, the demand for targeted therapies—such as nitrogen-scavenging agents, ammonia-lowering drugs, and emerging gene therapies—is expected to increase significantly.
Key industry players are investing heavily in research and development to advance innovative therapies. Strategic collaborations with regulatory bodies such as the U.S. FDA and European EMA are accelerating product approvals and facilitating faster market entry. Furthermore, companies are expanding global accessibility through strategic partnerships with healthcare providers and patient advocacy groups, particularly in underserved regions.
Awareness initiatives, early intervention programs, and advancements in metabolic monitoring are improving treatment adoption rates. As a result, the UCD treatment market value is projected to witness sustained expansion over the next decade.
→Market Overview
Urea Cycle Disorder (UCD) is a rare inherited metabolic condition characterized by the body’s inability to eliminate ammonia, a toxic by-product of protein metabolism. Deficiencies in any of the six urea cycle enzymes or two transporters lead to hyperammonemia, which can cause neurological damage, seizures, coma, or death if untreated.
Treatment strategies focus on reducing ammonia accumulation. Standard care includes:
- Low-protein dietary management supplemented with essential amino acids
- Nitrogen-scavenging medications such as sodium benzoate and sodium phenylbutyrate
- Glycerol phenylbutyrate formulations
- Dialysis in acute hyperammonemic crises
- Liver transplantation in severe or refractory cases
Long-term disease management requires strict ammonia monitoring, early intervention during metabolic crises, and multidisciplinary care approaches. Technological innovations in diagnostics and metabolic screening are enhancing early detection, thereby increasing therapeutic intervention rates.
→Key Market Growth Drivers
- Increase in Prevalence of Urea Cycle Disorder (UCD)
UCDs have an estimated annual incidence of approximately 1 in 35,000 births in the U.S., according to recent medical literature. Though classified as rare diseases, improved newborn screening programs and genetic testing technologies are increasing detection rates globally.
Growing awareness among clinicians, better access to metabolic testing, and expanded screening in emerging markets are contributing to rising diagnosed cases. This directly fuels demand for chronic treatment regimens and specialty drugs.
Furthermore, increased research funding for rare genetic disorders is enabling the development of novel therapeutic approaches such as mRNA-based gene therapy and enzyme-targeted treatments.
- Surge in Product Approvals
Regulatory agencies are prioritizing orphan diseases, granting fast-track designations and orphan drug benefits to companies developing UCD therapies. Accelerated approvals are increasing the number of available treatment options, enhancing patient outcomes.
New formulations, improved delivery mechanisms, and better tolerability profiles are encouraging broader adoption. As product pipelines mature, revenue opportunities are expected to expand steadily.
- Dominance of OTC Deficiency Segment
Ornithine Transcarbamylase (OTC) deficiency remains the most common and severe enzyme deficiency type within UCDs. The critical nature of OTC deficiency drives higher treatment penetration and continuous drug utilization.
Advancements in genetic therapy research targeting OTC deficiency are expected to contribute meaningfully to market growth during the forecast period.
- Hospital Pharmacies as Key Distribution Channel
Hospital pharmacies dominate distribution due to the specialized nature of UCD therapies. Acute hyperammonemia management often requires hospital-based intervention and intravenous administration, reinforcing hospital pharmacy leadership.
→Market Challenges & Opportunities
Challenges
- High treatment costs, particularly for gene therapies and enzyme replacement drugs
- Limited patient pool due to rarity of disease
- Complex clinical trial recruitment
- Stringent regulatory requirements
- Limited awareness in low-income regions
Opportunities
- Expansion of newborn screening programs globally
- Increasing orphan drug incentives
- Growing investment in gene-editing technologies
- Untapped markets in Asia Pacific and Latin America
- Strategic collaborations between biotech firms and global pharmaceutical companies
→Explore Strategies & Trends – Request Full Report Access – https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=86533
→Analysis of Key Players – Key Player Strategies
Leading companies operating in the global UCD treatment market include:
- Bausch Health Companies Inc.
- Eurocept Pharmaceutical Holding
- Zevra Therapeutics, Inc.
- Ultragenyx Pharmaceutical Inc
- Aeglea BioTherapeutics
- Arcturus Therapeutics, Inc.
- Orpharma Pty Ltd.
- Abbott
- Nestle SA
- Mead Johnson & Company, LLC
- Boehringer Ingelheim International GmbH
- CAMP4 Therapeutics
These companies are focusing on:
- Expanding orphan drug pipelines
- Investing in gene therapy and mRNA platforms
- Strengthening regional distribution networks
- Partnering with research institutions
- Enhancing product accessibility through differential pricing strategies
Biotechnology firms are particularly active in developing next-generation therapies targeting enzyme deficiencies at the genetic level, while established pharmaceutical players are leveraging global supply chains and regulatory expertise.
→Recent Developments
In September 2022, iECURE announced that the U.S. Food and Drug Administration (FDA) has granted Orphan Drug Designation to its lead product candidate GTP-506, an investigational product, for the treatment of Ornithine Transcarbamylase (OTC) deficiency, a rare genetic condition that can lead to irreversible neurological impairment, seizures, coma and death in a pediatric population.
In December 2022, Acer Therapeutics Inc. and its collaboration partner, RELIEF THERAPEUTICS Holding SA announced that the U.S. Food and Drug Administration (FDA) has approved OLPRUVA (sodium phenylbutyrate) for oral suspension in the U.S. for the treatment of certain patients living with urea cycle disorders (UCDs) involving deficiencies of carbamylphosphate synthetase (CPS), ornithine transcarbamylase (OTC), or argininosuccinic acid synthetase (AS).
→Investment Landscape and ROI Outlook
The UCD treatment market presents moderate but stable ROI potential due to:
- Recurring demand for lifelong therapies
- Orphan drug exclusivity benefits
- Premium pricing power
- Low competitive saturation compared to mainstream therapeutic categories
Investments in gene therapy platforms may offer higher long-term returns, particularly if one-time curative treatments replace chronic therapy models. Venture capital funding and strategic acquisitions are expected to remain active, particularly in North America and Europe.
Healthcare systems are increasingly recognizing the cost-benefit of early treatment to prevent neurological damage and intensive care admissions, strengthening reimbursement frameworks.
→Market Segmentations
By Treatment Type
- Glycerol Phenylbutyrate
- Amino Acid Supplements
- Sodium Phenylbutyrate
- Sodium Benzoate
- Others
By Enzyme Deficiency Type
- OTC deficiency
- ASL deficiency
- ASS1 deficiency
- CPSI deficiency
- NAGS deficiency
- ARG1 deficiency
- Others
By Route of Administration
- Oral
- Parenteral
By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
By Region
- North America (U.S., Canada)
- Europe (Germany, U.K., France, Italy, Spain)
- Asia Pacific (China, India, Japan, South Korea, Australia & New Zealand)
- Latin America (Brazil, Mexico)
- Middle East & Africa (GCC Countries, South Africa)
North America held the largest share in 2024 due to advanced healthcare infrastructure, favorable orphan drug policies, and strong R&D presence.
→Why Buy This Report?
- In-depth quantitative and qualitative analysis
- Detailed competitive landscape and company profiling
- Comprehensive regional and segment analysis
- Historical data (2020–2024) and forecast (2025–2035)
- Strategic insights for investors and stakeholders
- Coverage in Electronic (PDF) + Excel formats
- Customization available upon request
→FAQs
- How big was the global urea cycle disorder treatment market in 2024?
It was valued at US$ 527.5 Mn in 2024.
- How fast will the urea cycle disorder treatment industry grow during the forecast period?
The market is projected to grow at a CAGR of 3.5% from 2025 to 2035.
- What factors are driving the UCD treatment market?
Key drivers include increasing prevalence of UCDs, rising product approvals, expanding diagnostic capabilities, and growing orphan drug incentives.
- Which region dominates the global market?
North America is expected to account for the largest share during 2025–2035.
- Who are the prominent UCD treatment market providers?
Major players include Bausch Health Companies Inc., Eurocept Pharmaceutical Holding, Zevra Therapeutics, Inc., Ultragenyx Pharmaceutical Inc, Aeglea BioTherapeutics, Arcturus Therapeutics, Inc., Orpharma Pty Ltd., Abbott, Nestle SA, Mead Johnson & Company, LLC, Boehringer Ingelheim International GmbH., CAMP4 Therapeutics and other prominent players.
Buy Full Report Now: https://www.transparencymarketresearch.com/checkout.php?rep_id=86533<ype=S
→Explore Latest Research Reports by Transparency Market Research:
Contact and Intraocular Lenses Market: https://www.transparencymarketresearch.com/contact-and-intraocular-lenses-market.html
Foley Catheters Market: https://www.transparencymarketresearch.com/foley-catheter-market.html
Microcatheter Market: https://www.transparencymarketresearch.com/microcatheters-market.html
About Transparency Market Research
Transparency Market Research, a global market research company registered at Wilmington, Delaware, United States, provides custom research and consulting services. Our exclusive blend of quantitative forecasting and trends analysis provides forward-looking insights for thousands of decision makers. Our experienced team of Analysts, Researchers, and Consultants use proprietary data sources and various tools & techniques to gather and analyses information.
Our data repository is continuously updated and revised by a team of research experts, so that it always reflects the latest trends and information. With a broad research and analysis capability, Transparency Market Research employs rigorous primary and secondary research techniques in developing distinctive data sets and research material for business reports.


Leave a Reply