The global viral vector & plasmid DNA manufacturing market is witnessing rapid expansion, fueled by the growing adoption of gene therapies, cell-based treatments, and advanced biologics. Viral vectors and plasmid DNA are critical components in modern biotechnology, enabling the delivery of genetic material into cells for therapeutic and research purposes.
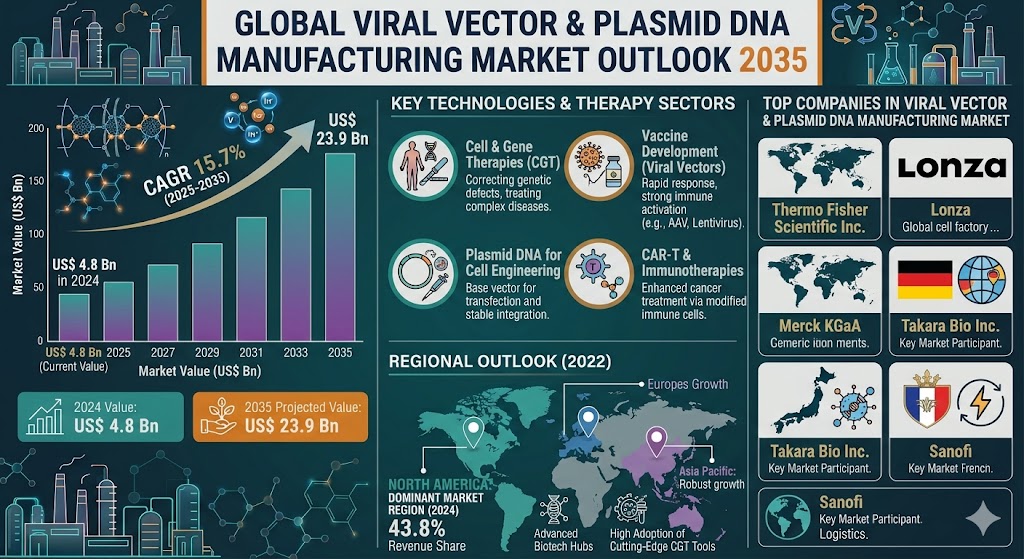
The global viral vector & plasmid DNA manufacturing market was valued at US$ 4.8 Billion in 2024 and is projected to reach US$ 23.9 Billion by the end of 2035, expanding at a compound annual growth rate (CAGR) of 15.7% from 2025 to 2035. The rising prevalence of genetic disorders, increasing clinical trials, and advancements in gene editing technologies are key factors driving market growth.
Market Overview
Viral vectors and plasmid DNA are essential tools used in gene therapy, vaccine development, and cell therapy applications. Viral vectors—such as adenoviral, lentiviral, and adeno-associated viral (AAV) vectors—are engineered to deliver therapeutic genes into target cells.
Plasmid DNA serves as a fundamental building block in the production of viral vectors and is also used directly in DNA vaccines and gene therapies. These materials are manufactured under strict quality standards to ensure safety, purity, and efficacy.
The growing focus on precision medicine and personalized therapies is significantly increasing the demand for high-quality viral vector and plasmid DNA manufacturing services.
Get your sample market research report copy today@ https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=30428
Rising Demand for Gene and Cell Therapies
One of the primary drivers of the market is the increasing adoption of gene and cell therapies for the treatment of complex and previously untreatable diseases. These therapies aim to address the root causes of diseases at the genetic level.
Viral vectors are widely used to deliver therapeutic genes in treatments for conditions such as cancer, rare genetic disorders, and neurological diseases. Similarly, plasmid DNA is essential in the development of CAR-T cell therapies and DNA-based vaccines.
Expansion of Biopharmaceutical R&D and Clinical Trials
The biopharmaceutical industry is investing heavily in research and development (R&D) to develop innovative therapies. The increasing number of clinical trials involving gene therapies and biologics is driving the need for scalable and reliable manufacturing solutions.
Contract development and manufacturing organizations (CDMOs) are playing a crucial role in meeting this demand by providing specialized manufacturing services. These organizations offer expertise, infrastructure, and regulatory compliance, enabling faster development and commercialization of therapies.
Technological Advancements in Manufacturing Processes
Advancements in manufacturing technologies are improving the efficiency, scalability, and quality of viral vector and plasmid DNA production. Innovations such as suspension cell culture systems, single-use bioreactors, and continuous manufacturing processes are enhancing production capabilities.
Automation and digital monitoring systems are also being integrated into manufacturing workflows to ensure consistency and reduce production costs. These advancements are enabling manufacturers to meet the increasing demand for high-quality genetic materials.
Increasing Demand for Vaccines and Advanced Therapeutics
The demand for vaccines, particularly DNA and RNA-based vaccines, has increased significantly in recent years. Viral vectors and plasmid DNA are key components in vaccine development and production.
The success of advanced vaccine platforms has accelerated research in this field, leading to increased investments in manufacturing infrastructure. The growing focus on pandemic preparedness and rapid vaccine development is expected to drive further demand.
Key Developments in Viral Vector & Plasmid DNA Manufacturing Market
- In June 2025, ProBio announced the opening of its Cell and Gene Therapy Center of Excellence at Princeton West Innovation Campus in Hopewell, New Jersey. The 128,000 sq. ft. state-of-the-art GMP facility is equipped with a view to high-quality plasmid DNA and viral vector manufacturing, including adeno-associated virus (AAV) and lentiviral vector (LVV) platforms, highlighting ProBio’s focus on driving the accelerated delivery of life-changing medicines.
- In June 2025, ArcticZymes Technologies ASA announced the expansion of its GMP product line, with M-SAN HQ GMP, a new GMP grade nuclease for use in virus vector manufacturing. The newly launched product brought the established performance of M-SAN HQ with regulatory strength of GMP manufacturing and offered an extremely efficient enzymatic host cell DNA elimination solution under physiological salt conditions.
Get this premium report for strategic insights: https://www.transparencymarketresearch.com/checkout.php?rep_id=30428<ype=S
Regional Analysis
North America
North America dominates the global viral vector & plasmid DNA manufacturing market due to its strong biotechnology ecosystem and advanced healthcare infrastructure. The United States is a major contributor, with a high concentration of biopharmaceutical companies, research institutions, and CDMOs.
Europe
Europe is another key market, driven by increasing research activities and government support for biotechnology innovation. Countries such as Germany, the United Kingdom, and Switzerland are actively investing in gene therapy development and manufacturing capabilities.
Asia Pacific
Asia Pacific is expected to witness the fastest growth during the forecast period due to expanding biotechnology industries and increasing healthcare investments. Countries such as China, India, Japan, and South Korea are strengthening their capabilities in biologics manufacturing and gene therapy research.
Latin America and Middle East & Africa
These regions are gradually emerging as potential markets due to improving healthcare infrastructure and increasing awareness of advanced therapies. Investments in biotechnology and pharmaceutical sectors are expected to drive future growth.
Key Companies in the Viral Vector & Plasmid DNA Manufacturing Market
The global market is highly competitive, with several companies focusing on expanding production capacity and enhancing technological capabilities. Key players include:
- Thermo Fisher Scientific Inc.
- Lonza
- Hillgene Biopharma Co., Ltd.
- Revvity
- Merck KGaA
- Aldevron LLC
- VGXI
- FUJIFILM Biotechnologies
- WuXi Biologics
- Takara Bio Inc.
- REGENXBIO Inc.
- AGC Biologics
- Batavia Biosciences B.V.
- Sanofi
- Wacker Chemie AG
These companies are investing in advanced manufacturing technologies and strategic collaborations to strengthen their market position.
Market Challenges
Despite strong growth potential, the market faces challenges such as high production costs, complex manufacturing processes, and stringent regulatory requirements. Limited manufacturing capacity and scalability issues can also impact the timely delivery of therapies.
Additionally, maintaining product quality and consistency is critical, requiring significant investment in infrastructure and expertise.
Future Outlook
The global viral vector & plasmid DNA manufacturing market is poised for substantial growth as the adoption of gene therapies and advanced biologics continues to rise. Technological advancements, increasing R&D investments, and expanding clinical pipelines will drive market expansion.
With the market projected to reach US$ 23.9 Billion by 2035, viral vector and plasmid DNA manufacturing will remain a cornerstone of next-generation therapeutics and precision medicine.
Frequently Asked Questions (FAQs)
Q1. What is the projected size of the viral vector & plasmid DNA manufacturing market by 2035?
The market is expected to reach US$ 23.9 Billion by the end of 2035.
Q2. What is the expected CAGR of the market?
The market is projected to grow at a CAGR of 15.7% from 2025 to 2035.
Q3. What are the key drivers of the market?
Key drivers include rising demand for gene and cell therapies, increasing clinical trials, technological advancements, and growing investments in biotechnology.
Q4. Which region dominates the market?
North America dominates due to strong biotechnology infrastructure and high R&D investments.
Q5. What are the main applications of viral vectors and plasmid DNA?
They are primarily used in gene therapy, vaccine development, cell therapy, and advanced biologics manufacturing.
Explore Latest Research Reports by Transparency Market Research:
Flooring Market : https://www.transparencymarketresearch.com/flooring-market.html
Produced Water Treatment Market: https://www.transparencymarketresearch.com/produced-water-treatment-market.html
Artificial Intelligence Chipset Market: https://www.transparencymarketresearch.com/artificial-intelligence-chipsets-market.html
Refurbished Computers and Laptops Market: https://www.transparencymarketresearch.com/refurbished-computers-laptops-market.html
About Transparency Market Research
Transparency Market Research, a global market research company registered at Wilmington, Delaware, United States, provides custom research and consulting services. Our exclusive blend of quantitative forecasting and trends analysis provides forward-looking insights for thousands of decision makers. Our experienced team of Analysts, Researchers, and Consultants use proprietary data sources and various tools & techniques to gather and analyses information.
Our data repository is continuously updated and revised by a team of research experts, so that it always reflects the latest trends and information. With a broad research and analysis capability, Transparency Market Research employs rigorous primary and secondary research techniques in developing distinctive data sets and research material for business reports.
Contact Us:
Transparency Market Research Inc.
CORPORATE HEADQUARTER DOWNTOWN,
1000 N. West Street,
Suite 1200, Wilmington, Delaware 19801 USA
Tel: +1-518-618-1030
USA – Canada Toll Free: 866-552-3453


Leave a Reply